By ICS International Certification LLP
Selecting the right supplier is one of the most critical decisions for any organization. A poor choice can lead to quality failures, delayed deliveries, increased costs, and reputational damage. On the other hand, a well-evaluated supplier becomes a long-term partner in growth and reliability.
This guide outlines a structured and practical approach to evaluating a new supplier before starting production.
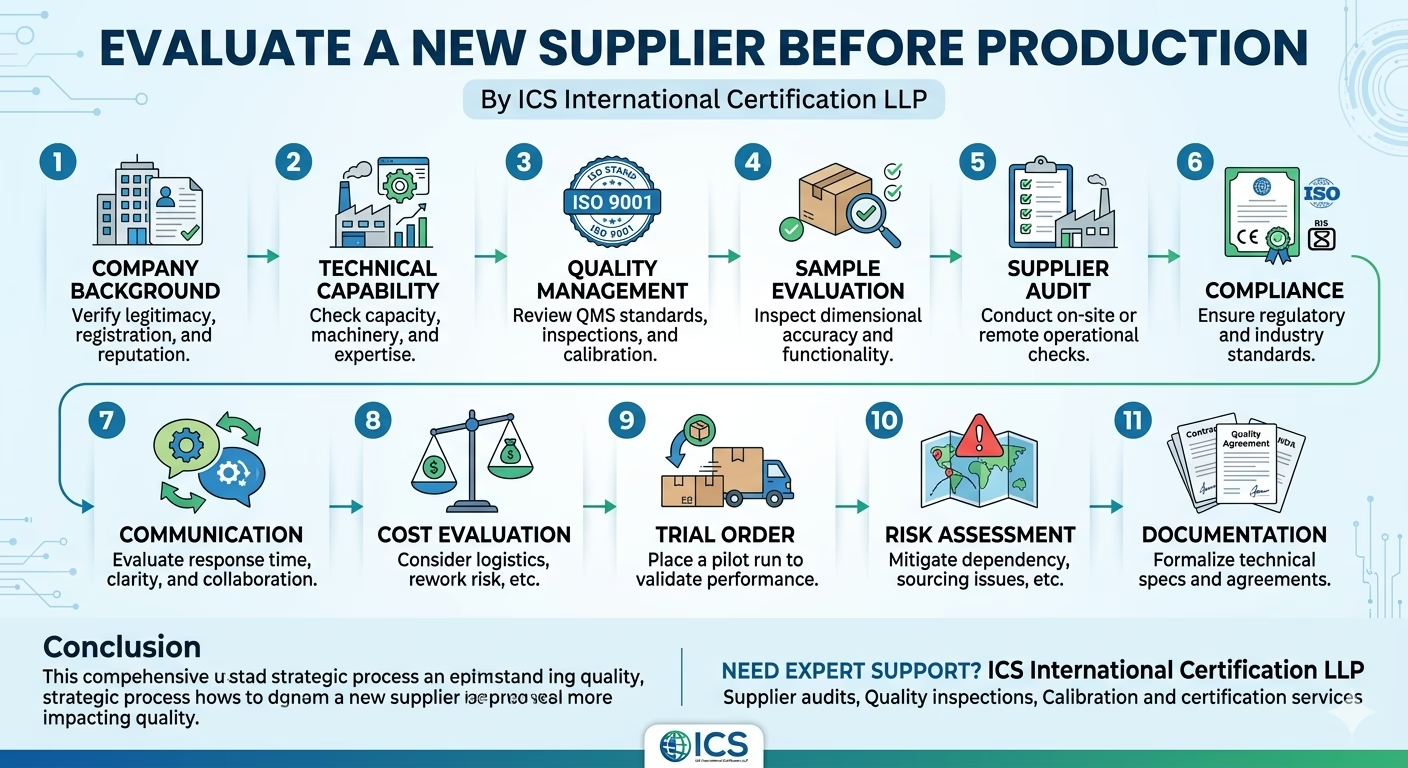
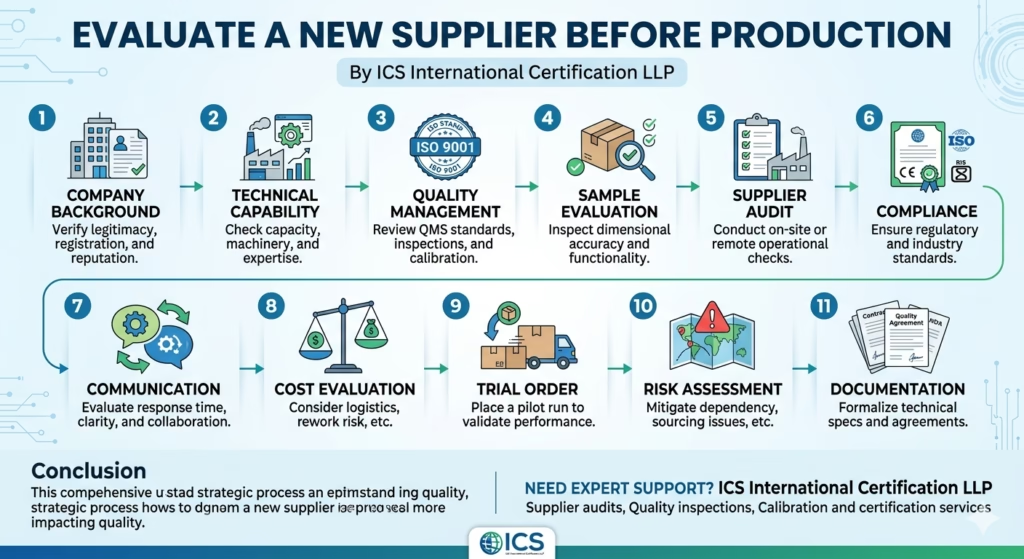
1. Company Background Verification
Before engaging with any supplier, verify their legitimacy and stability. This includes checking business registration, certifications, years of operation, and market reputation.
A reliable supplier should be transparent in sharing:
- Company registration documents
- Certification details (ISO, industry-specific)
- Client references
This step ensures you are dealing with a credible and established organization.
2. Technical Capability Assessment
A supplier must have the technical ability to meet your requirements consistently.
Key aspects to evaluate:
- Production capacity and scalability
- Machinery and technology used
- Skilled workforce and technical expertise
- Experience with similar products or industries
Matching supplier capability with your product requirements is essential to avoid production challenges later.
3. Quality Management System (QMS) Review
A strong quality system is the backbone of consistent production.
Look for:
- Implementation of ISO 9001 or equivalent standards
- Defined inspection procedures (incoming, in-process, final)
- Calibration of measuring instruments
- Traceability and documentation systems
A well-implemented QMS ensures that quality is built into the process, not just inspected at the end.
4. Sample Evaluation
Always request samples before finalizing a supplier.
Evaluate samples for:
- Dimensional accuracy
- Material compliance
- Surface finish
- Functional performance
Where required, samples should be tested through internal labs or third-party inspection agencies to validate compliance with specifications.
5. Supplier Audit (On-site or Remote)
Conducting a supplier audit provides deeper insight into their operations.
Areas to assess:
- Manufacturing processes and workflow
- Quality control checkpoints
- Storage and material handling practices
- Cleanliness and workplace organization
A structured audit helps identify potential risks before they impact production.
6. Compliance and Certifications
Ensure the supplier meets all relevant regulatory and industry requirements.
Depending on your industry, this may include:
- ISO certifications (9001, 14001, etc.)
- Product certifications (CE, BIS, FDA, etc.)
- Social and ethical compliance standards
Compliance is not just a requirement—it reflects the supplier’s commitment to quality and responsibility.
7. Communication and Responsiveness
Effective communication is often underestimated but crucial.
Evaluate:
- Response time and clarity
- Technical understanding
- Problem-solving approach
- Willingness to collaborate
Strong communication ensures smoother coordination and faster resolution of issues.
8. Cost Evaluation Beyond Price
Choosing a supplier based solely on price can be risky.
Consider the total cost of ownership, including:
- Tooling and setup costs
- Logistics and shipping
- Payment terms
- Risk of rework, rejection, or delays
A slightly higher price with better reliability often results in lower overall costs.
9. Trial Order / Pilot Run
Before moving to full-scale production, place a trial order.
Monitor:
- Delivery timelines
- Product consistency
- Packaging quality
- Responsiveness during execution
This step helps validate real-world performance.
10. Risk Assessment
Identify and mitigate potential risks associated with the supplier.
Common risks include:
- Dependency on a single supplier
- Raw material sourcing issues
- Geographic or political risks
- Capacity constraints
Having contingency plans ensures business continuity.
11. Documentation and Agreements
Formalize the relationship with proper documentation.
This should include:
- Technical specifications
- Quality agreements
- Non-disclosure agreements (if required)
- Penalty clauses for delays or non-conformance
Clear documentation minimizes misunderstandings and protects both parties.
Conclusion
Evaluating a new supplier is not just a one-time activity—it is a strategic process that directly impacts product quality, operational efficiency, and customer satisfaction.
At ICS International Certification LLP, we emphasize structured supplier evaluation as a key element of quality assurance and risk management. A well-chosen supplier is not just a vendor, but a trusted partner in your organization’s success.
Need Expert Support?
ICS International Certification LLP offers professional services including:
- Supplier audits
- Quality inspections
- Calibration and certification services
Contact us to ensure your supplier selection process is robust, reliable, and compliant with international standards.