STANDARD OPERATING PROCEDURE (SOP)
Control of Non-Conforming Products After Inspection
Issued By: ICS INTERNATIONAL CERTIFICATION LLP
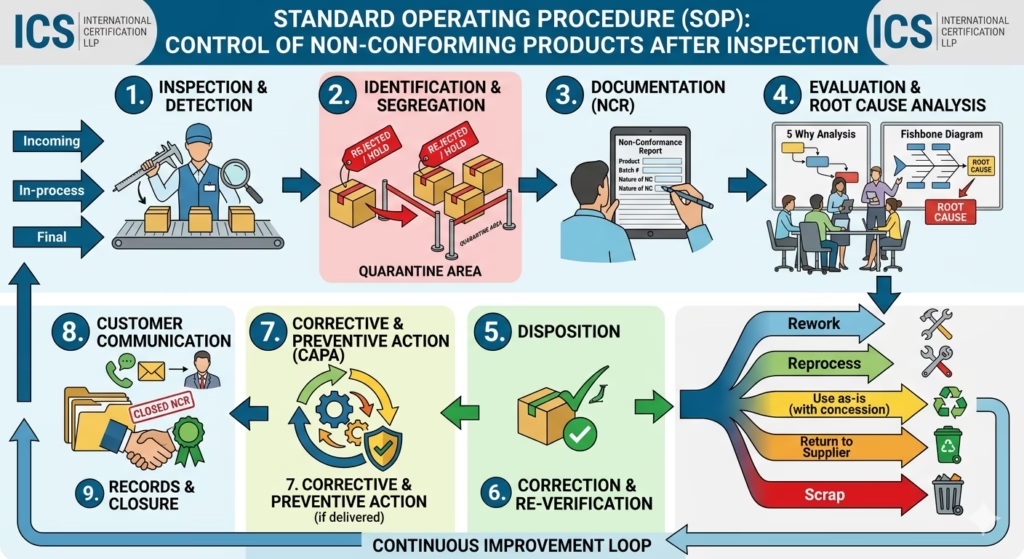
1. Purpose
To establish a systematic process for identification, control, and disposition of non-conforming products to prevent unintended use or delivery.
2. Scope
This procedure applies to all products inspected at:
- Incoming stage
- In-process stage
- Final inspection stage
3. Responsibility
- Inspector / Quality Control (QC): Identification and reporting
- Quality Manager: Review and disposition approval
- Production Team: Rework / correction
- Top Management (if required): Approval for concession
4. Procedure
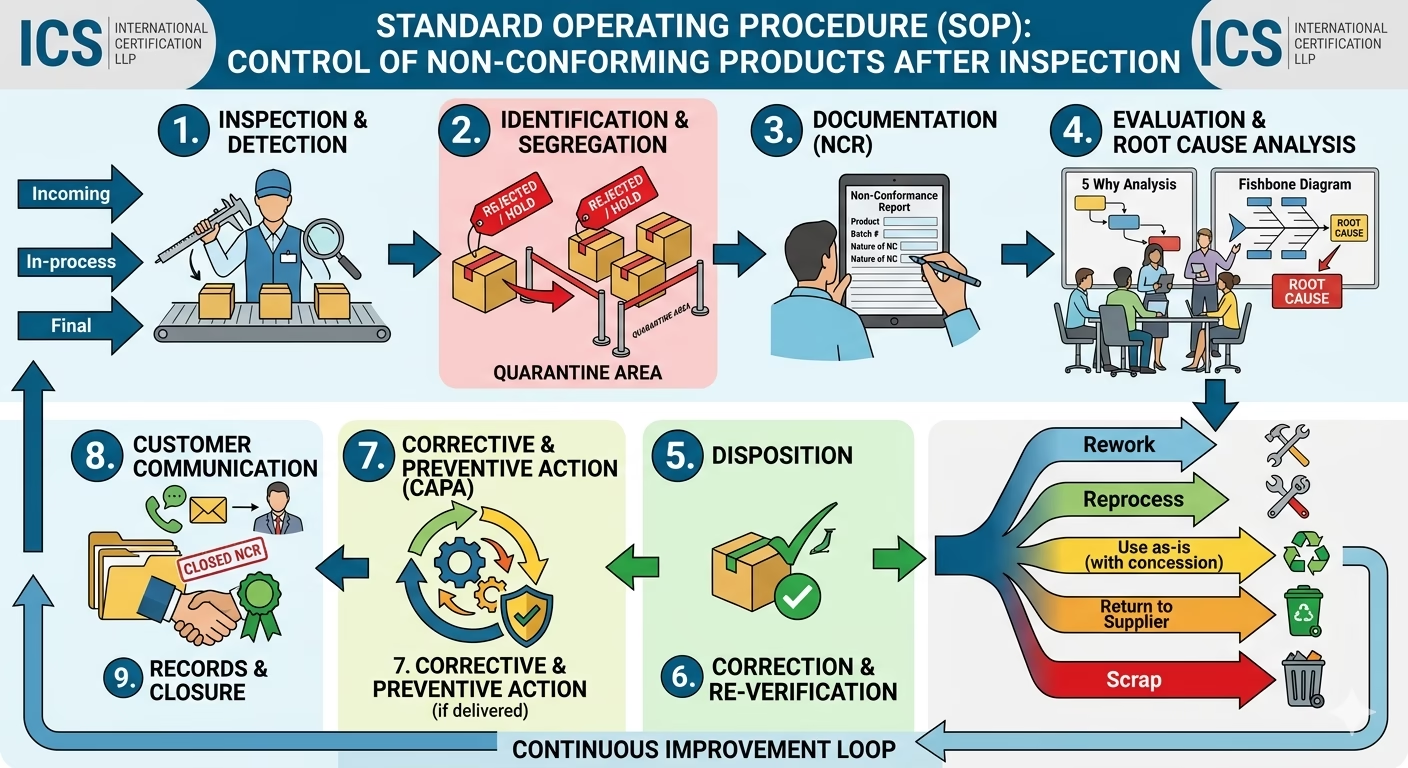
4.1 Identification of Non-Conforming Product
- Products failing to meet specified requirements shall be identified during inspection.
- Non-conformity may include:
- Dimensional deviation
- Visual defects
- Functional failure
4.2 Segregation
- Non-conforming products shall be:
- Clearly marked as “REJECTED / HOLD”
- Physically segregated in a designated Quarantine Area
4.3 Documentation (NCR)
- A Non-Conformance Report (NCR) shall be raised including:
- Product details
- Batch / Lot number
- Nature of non-conformity
- Quantity affected
- Inspection details
4.4 Evaluation & Root Cause Analysis
- The Quality team shall analyze the cause using:
- 5 Why Analysis
- Fishbone Diagram
- Root cause must be identified before disposition.
4.5 Disposition of Non-Conforming Product
The product shall be reviewed and one of the following actions taken:
- Rework: Correct the defect
- Reprocess: Repeat the process
- Use as-is: With documented approval (concession)
- Return to Supplier: If supplier-related issue
- Scrap: If unusable
All decisions must be approved by authorized personnel.
4.6 Correction & Re-Verification
- Reworked or reprocessed products shall be re-inspected.
- Only conforming products shall be released.
4.7 Corrective and Preventive Action (CAPA)
- Corrective actions shall be implemented to eliminate root causes.
- Preventive measures shall be defined to avoid recurrence.
4.8 Customer Communication
- If non-conforming product is already delivered:
- Customer shall be informed immediately
- Necessary actions shall be taken (recall / replacement)
4.9 Records & Closure
Maintain records of:
- NCR
- Root Cause Analysis
- Disposition decision
- CAPA actions
- Final approval
All records shall be retained as per document control requirements.
5. Reference
- ISO 9001:2015 – Clause 8.7 (Control of Non-Conforming Outputs)
6. Process Flow
Inspection → Identification → Segregation → NCR → Analysis → Disposition → Correction → Re-Inspection → Closure
7. Approval
| Prepared By | Reviewed By | Approved By |
|---|---|---|
| Quality Team | Quality Manager | Top Management |
© ICS INTERNATIONAL CERTIFICATION LLP – All Rights Reserved