By ICS International Certification LLP
In today’s competitive industrial environment, maintaining consistent product quality is critical for operational excellence and customer satisfaction. As a trusted provider of certification, inspection, and compliance solutions, ICS International Certification LLP emphasizes the implementation of Quality Gates as a fundamental practice in manufacturing inspection systems.
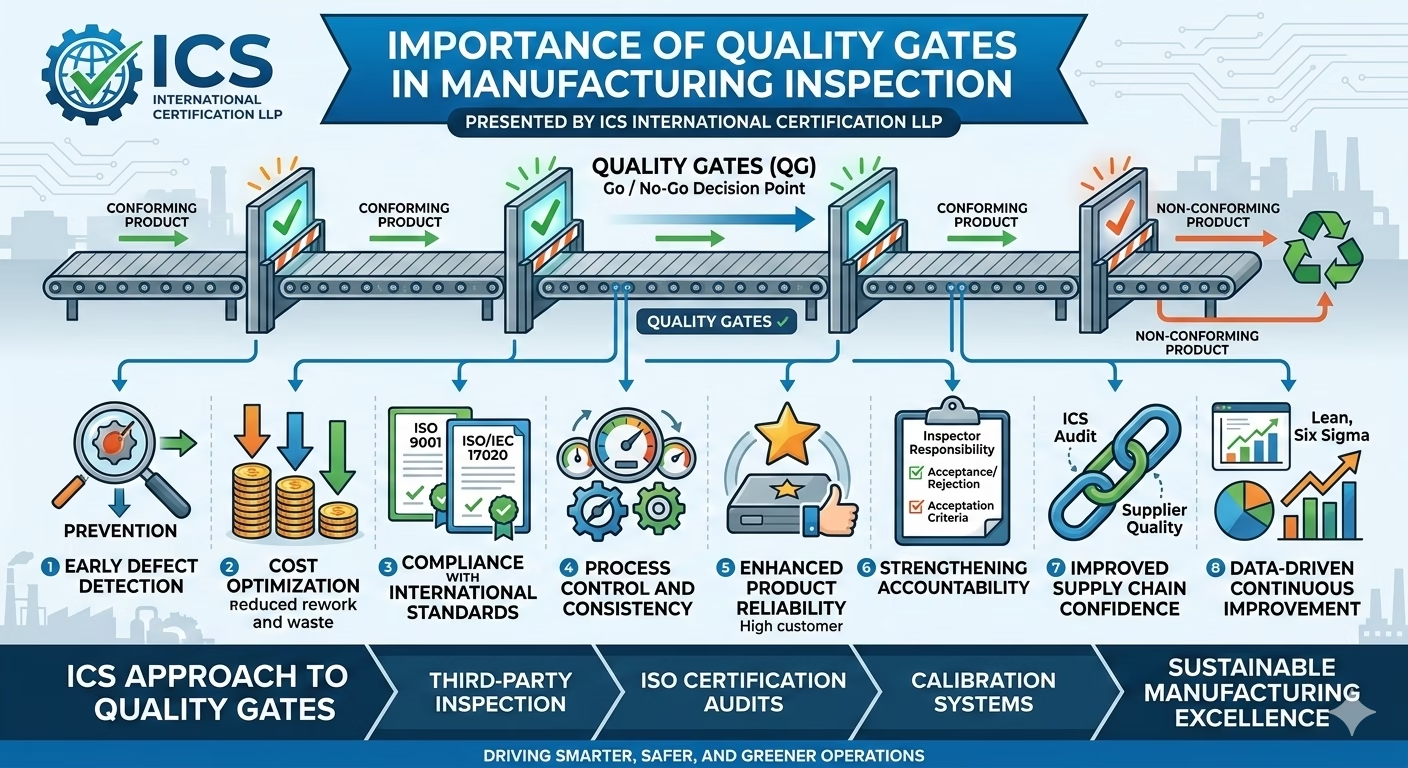
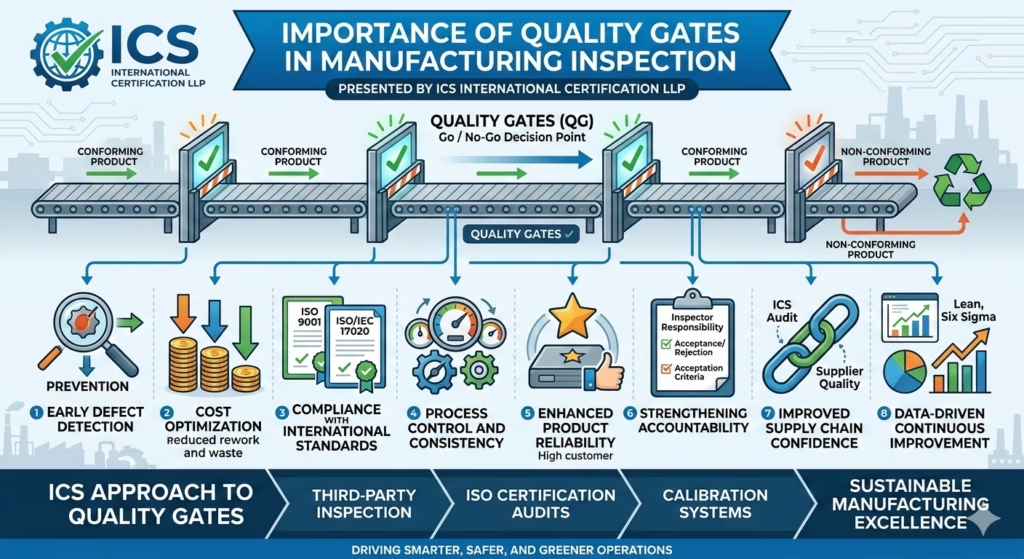
🔍 What Are Quality Gates?
Quality gates are defined inspection checkpoints within the manufacturing process where products are evaluated against specified standards before proceeding to the next stage.
They act as decision points (Go / No-Go) to ensure only conforming products move forward.
🎯 Why Quality Gates Are Important
1. Early Defect Detection
Quality gates enable identification of defects at the earliest stage, preventing defective components from progressing further in production.
➡ This aligns with ICS’s focus on preventive quality assurance rather than corrective actions.
2. Cost Optimization
By stopping defects early:
- Rework and scrap costs are minimized
- Resource wastage is reduced
- Overall production cost efficiency improves
3. Compliance with International Standards
Quality gates support adherence to standards such as:
- ISO 9001 (Quality Management Systems)
- ISO/IEC 17020 (Inspection bodies)
ICS International Certification LLP provides inspection services aligned with such global frameworks, ensuring regulatory compliance and audit readiness. (ICS International Certification)
4. Process Control and Consistency
They ensure:
- Standardized inspection criteria
- Reduced process variation
- Stable and repeatable manufacturing outputs
5. Enhanced Product Reliability
Only verified and conforming products pass each stage, resulting in:
- Improved product performance
- Reduced field failures
- Higher customer confidence
6. Strengthening Accountability
Each quality gate defines:
- Responsibility of inspectors or departments
- Clear acceptance/rejection criteria
This improves traceability and simplifies root cause analysis.
7. Improved Supply Chain Confidence
As industries face increasing supply chain pressure, structured inspection systems like quality gates ensure suppliers maintain consistent quality levels—an area where ICS actively supports organizations through second-party audits and inspections. (ICSIC)
8. Data-Driven Continuous Improvement
Quality gate records provide valuable data to:
- Identify recurring defects
- Improve processes
- Support Lean, Six Sigma, and operational excellence initiatives
🏭 ICS Approach to Quality Gates
ICS International Certification LLP integrates quality gates within its:
- Third-party inspection services
- ISO certification audits
- Calibration and compliance systems
This integrated model helps industries achieve “Smarter, Safer, and Greener” operations through structured quality control and continuous improvement. (Hindustan Bytes)
✅ Conclusion
Quality gates are not just inspection checkpoints—they are strategic control mechanisms that:
- Prevent defects
- Reduce costs
- Ensure compliance
- Enhance product quality
With the expertise of ICS International Certification LLP, organizations can implement robust quality gate systems that align with international standards and drive sustainable manufacturing excellence.